Osaka University
Graduate School of Engineering Science
Kawano
Laboratory
Hydrogen Storage
In recent, carbon materials are known to be effective for hydrogen storage.
However, the detailed mechanisms have not been clarified yet.
We performed computer simulations for hydrogen adsorption on graphene surfaces as shown by the figure below.
As a result, it was clarified that the wavefunction (shown by green- and red-colored regions) on H2 transfers to the graphene
as the H2 molecule approaches the graphene surface and that dissociative adsorption of H2 is enhanced near the surface.[1]
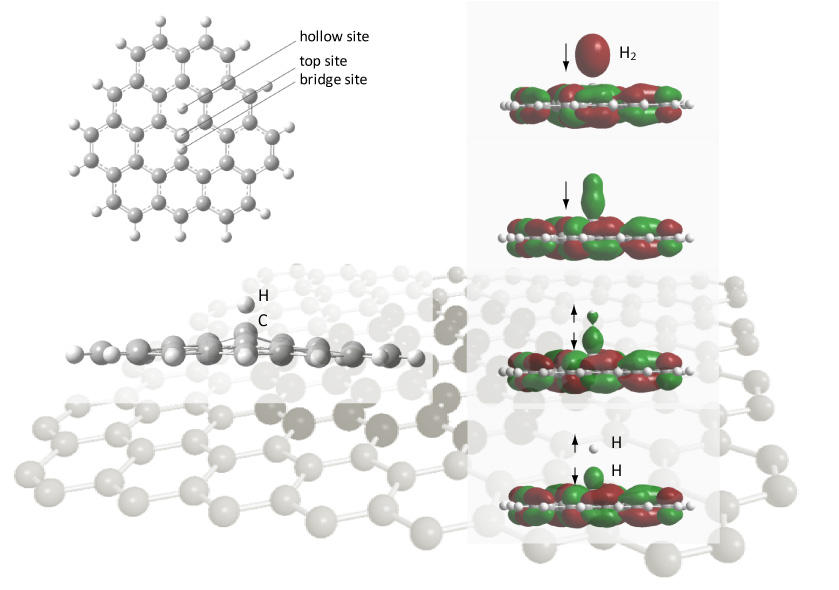 |
| Figure 1 Dissociative adsorption of H2 molecule near graphene surface. |
Li-ion Battery and Fuel Cell
Li-ion batteries and fuel cells have achieved impressive growth as one of leading technologies.
We focus on them and are trying to clarify the detailed mechanism of phenomena observed at the electrodes,
electrolytes, and their interfaces from the viewpoint of molecular scale.
In our own model, ion flow dynamics are treated on the basis of the multi-phase flow theory. [2]
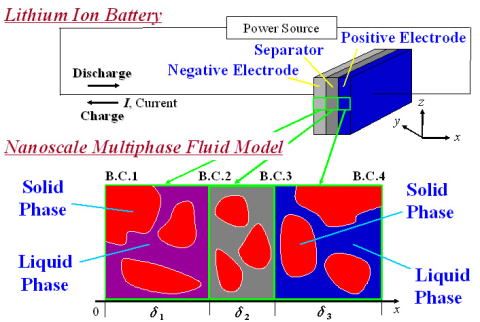 |
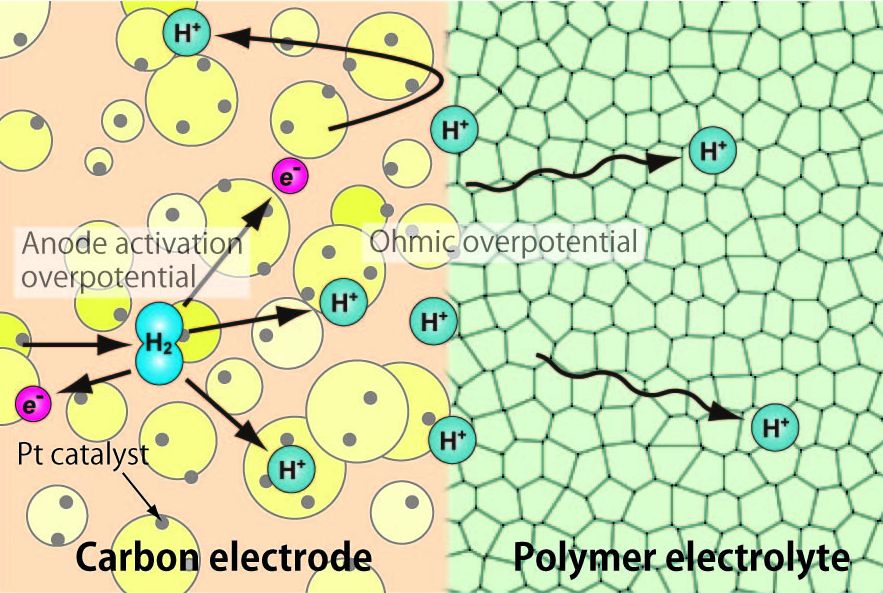 |
| Figure 2 Theoretical model of Li-ion battery. | Figure 3 Schematic of proton flow in PEFC. |
| You can see details in: |
| [1] Kentaro DOI, Ikumi ONISHI, and Satoyuki KAWANO, Ab initio Molecular Dynamics of H2 Dissociative Adsorption on Graphene Surfaces, Computer Modeling in Engineering & Sciences, Vol. 77, No. 2 (2011), pp. 113-136. |
| [2] Satoyuki KAWANO and Futoshi NISHIMURA, Numerical Analysis of Discharge Characteristics in Lithium Ion Batteries Using Multiphase Fluids Model, Jpn. J. Appl. Phys., Vol. 44, No. 6A (2005), pp. 4218-4228. |